Scientists:
Eva Kudová (PI), Ewa Szczurowska, Jiří Paleček, Jan Jakubík
Challenge
Neuropathic pain remains a therapeutic area with unmet medical needs. There is about 7% of the population suffering from neuropathy caused by diabetes, chemotherapy, ischemia, spinal cord injury or herpes. According to the clinical evidence, 40% of those people get only partial pain relief from available medicaments like analgesics, Lyrica, duloxetine, opioids or Qutenza.
Searching for novel drugs potentially useful for therapy of CNS damage belongs to the most investigated topics in contemporary pharmacology and neuroscience. The proposed project is based on the study of biological properties of newly developed 3αC substituted derivatives of pregnanolone and the evaluation of their pharmacotherapeutic potential. The current medical treatment of the Neuropathic Pain relies on the ion channel blockers (Gabapentin, Pregabalin) or the serotonin up-take blockers (Duloxetine) or the NMDA antagonists (Memantine, Dextromethorphan). Since direct antagonists or agonists or channel blockers can also severely influence physiological functions, they do have serious adverse side effects during the treatment. Our approach is focused on the allosteric modulation of the neuro-receptors, which is milder and only changes the sensitivity of the neuro-receptors to endogenous neurotransmitters or agonists.
Technology
The new proprietary steroids have proven their efficacy in increasing the reaction threshold to mechanical and thermal stimuli in six different animal models in rat.
The development candidate MS-225 shows good efficacy at doses of 1-3 mg/kg in comparison to the positive control by Gabapentin at 50-100 mg/kg. There were no obvious adverse effects observed up to 10 mg/kg. The MS-225 is bioavailable after p.o. administration and penetrates the BBB.
The neurosteroids act as positive allosteric modulators neuro-receptors.
Application of MS-225 significantly reduced mechanical allodynia in the Bortezomib induced peripheral neuropathy (BIPN) model. The effects were comparable to Gabapentin:
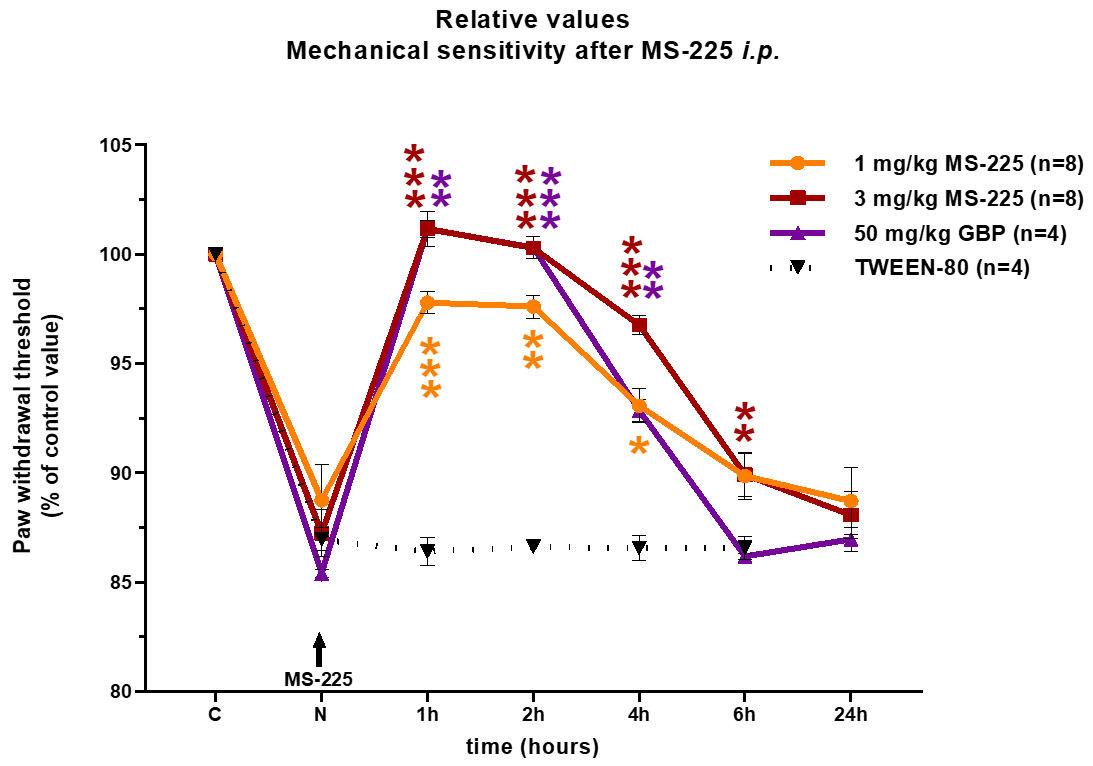
- Experiments were performed only after neuropathy (N) was fully established and confirmed by behavioural tests 2-3 weeks after the first Bortezomib administration
- MS-225 (1 and 3 mg/kg dose, i.p.), Gabapentin (50 mg/kg, i.p.), TWEEN-80 (PBS + 1% Tween-80, i.p.)
- *P<0.05, **P<0.01, ***P<0.001, when compared to the neuropathy value (N) for each group, One Way RM ANOVA, Dunnett’s post hoc test
- Abbreviations: C-control value, N-Neuropathy, an arrow indicates the time of MS-225 injection, GBP – Gabapentin
MS-225 intrathecal application potentiates the analgesic effects of endomorphin-1 in thermal sensitivity test in naïve rats:
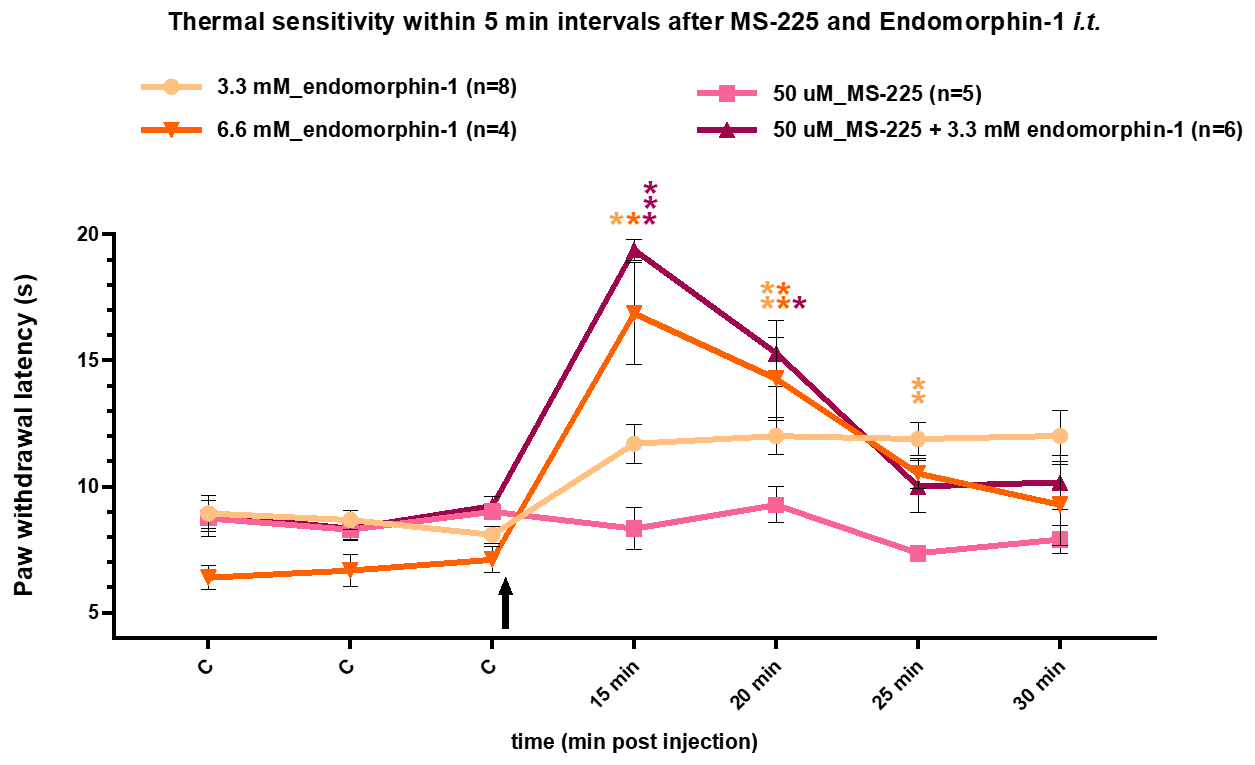
- Naïve animals with implanted intrathecal catheter were injected i.t. with 10𝜇l of 3,3mM (20𝜇g) or 6.6mM (40𝜇g) endomorphin-1 solution in 0.1% BSA , 10𝜇l of 50𝜇M (0.187𝜇g) MS-225 solution in 0,1% DMSO, or combination of endomorphin + MS-225.
- Thermal sensitivity test (Hargreaves Test) started 15 min after the administration
- 20s of exposure to high temperature is the test cut-off time to prevent thermal injury.
- With the endomorphin-1 + MS-225 combination several tested animals reached the cut-off value due to a strong analgesic effect.
- Endomorphin-1 at the higher dose of 40𝜇g (6.6mM) caused rapid but transient bilateral hind limb motor deficits lasting up to 10 min post injection. This effect was not present for the MS-225 and the M-S225 endomorphin-1 (3.3mM (20𝜇g)) combination.
- Abbreviations: C-control values; an arrow indicates time of intrathecal injections
Commercial opportunity
The Neuropathic Pain market is currently valued at USD 5 billion with a growing tendency to 10 bn. in 2028. A new efficacious small molecule with low or negligible side effects can easily acquire 30% of the market.
Development status
Preclinical development, lead optimisation, target validation
Categories
Neurosteroids; Positive Allosteric Modulators (PAM) of neuroreceptors
Keywords
neuropathic pain opioid receptors steroids pain relief neurosteroids allosteric modulation